Breathable air in a refuge chamber is the air that can support life. It has enough oxygen in its mix and doesn’t contain contaminants like carbon monoxide, carbon dioxide, water, or other toxins that will poison refuge chamber occupants during entrapment.
Multiple breathable air sources within a refuge station need to be supplied to accommodate various durations and events.
Sources of Breathable Air in a Refuge Chamber
A refuge chamber is essentially an airtight structure that restricts natural air from entering the internal atmosphere; without an additional air supply, occupants would quickly suffocate. Rough calculations show this would occur within 2-3 hours.
The Western Australian Department of Mines and Petroleum’s Guidelines on Refuge Chambers in Underground Mines’ stipulates a minimum of two independent sources of breathable air are to be supplied to a refuge chamber. MineARC Systems recommends a third source of breathable air wherever possible.
The two standard sources of breathable air within a refuge chamber are:
Incoming, filtered, and regulated compressed air from an external source.
Usually, this is a large surface compressor.
A scrubbing system used in conjunction with oxygen cylinders.
The scrubbing system removes toxins (carbon dioxide and carbon monoxide), and the oxygen bottles reintroduce oxygen at the rate it is being consumed.
The third source is optional but recommended; chemically produced oxygen from an oxygen candle.
O2 candles are used in an extreme emergency, in the event of failure of the first two systems, to either extend the duration or to buffer a response time to exit the chamber.
Access More Resources

Compressed Air to a Refuge Bay
Compressed air is simply the atmospheric “air” from above being pushed through pipelines down to the refuge chamber via a compressor.
A regulator adjusts the flow rate of the compressed air into the emergency refuge chamber.
The flow of compressed air required to sustain a breathable atmosphere within a safe refuge chamber is calculated at;
- 85L/m (3CFM) for the shelter, and
- 85L/m (3CFM) per person after that.
Compressed Air Filtration
Before compressed air can be considered suitable for breathing, it needs to be filtered from the following contaminants:
- Water that has been compressed from humidity in the air (airborne water particles). If water is not removed, it will increase the humidity within the refuge chamber to the point where the heat index increases to an uninhabitable level.
- Oil (as a liquid), drawn from the lubricants of the mechanical elements in the compressor, or smoke from oil or lubricants that are burnt during the compressing process. Oil mist can cause breathing discomfort as well as sickness and pneumonia.
- Pollutants such as carbon monoxide and hydrocarbons from diesel exhausts aren’t uncommon in a compressor’s airline. Carbon monoxide only needs to exist at 30 parts per million to cause physical effects. Also diesel particulate can cause breathing discomfort and irreversible lung damage.
Compressed Air Filtration Order
The order of a filtration system’s elements is critical in ensuring the compressed air supplied to a refuge chamber is safe for occupants to breathe.
The MineARC Compressed Air Management System (CAMS) is tested to ISO 12500-1 and has been specifically designed to meet AS/NZS 1716:2003.
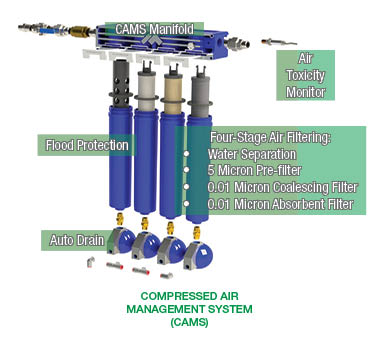
The CAMS flood protector prevents large amounts of water from passing any further through the filtration system. In contrast, the centripetal water separator spins smaller water droplets out of the air and lets them collect in the bottom of the filter housing to be disposed of via the auto drain.
The pre-filter is specifically designed to remove large particles of water and oil from the incoming compressed air source.
The element materials are galvanised steel inner and outer sleeved tubes, which create turbulence without a significant pressure drop. Micro glass fabric sheets are positioned between these tubes.
The coalescent filter removes the finer particles of oil and water from the incoming compressed air.
A borosilicate micro-glass element can absorb both water and oil particles, while a glass scrim allows the water to flow to the auto drain.

Chemical Scrubbing & Oxygen Cylinders
If the primary, compressed air source fails, the refuge chamber must have an independent source of oxygen and remove toxins to keep the atmosphere inhabitable.
Combining compressed oxygen cylinders and a chemical scrubbing system to filter the air of carbon dioxide (CO2) and carbon monoxide (CO) will achieve breathable air.
Compressed Oxygen Cylinders
A regulator controls the flow rate of compressed oxygen released into the chamber. Set the flow at the rate of occupant consumption, which is 0.5-0.6L per person/per minute.
MineARC recommends using the largest medical grade oxygen cylinder – ‘G size’ – containing 8200L of breathable oxygen.
CO and CO2 Chemical Scrubber
Air scrubbing is the process of chemically removing contaminants or undesired gases from the air. Scrubbing differs from filtration in that a filter is a mesh that screens the air and captures solid particles. In contrast, scrubbing uses chemical reactions to change the composition of gases as they pass through the system.
In any sealed environment, carbon monoxide and carbon dioxide can build up and poison the air. In high enough concentrations, both CO2 and CO can cause serious harm to occupants. Hence, the removal of these gases is vital for any refuge shelter.
Removing Carbon Dioxide Inside a Refuge Bay
Inside a MineARC refuge chamber, the scrubbing systems can absorb 1400L of carbon dioxide using a soda-lime product which generates a chemical reaction converting CO2 into an inert substance.
Each occupant produces 30.6L/hour of CO2, which will need to be removed by recirculating the chamber’s air through the chemical bed. As the chemical reaction comes to an end, the cartridge is changed out and starts again.
Removing Carbon Monoxide Inside a Refuge Bay
Carbon monoxide is scrubbed from the air using a catalytic process. The reaction is exothermic, which means it generates heat as a by-product.
Air is passed through the scrubber chemical, converting carbon monoxide into CO2 and H2O. The carbon dioxide produced is then removed by soda-lime.
Only one CO cartridge is required per portable refuge chamber. The cartridges are also effective at removing other gases, such as ethylene oxide, hydrogen and ethane.
Emergency Oxygen Candles
The oxygen candle is the third and final breathable air supply method. The candle uses a chemical reaction to generate oxygen.
Oxygen candles burn for approximately 60-90 minutes, producing 2600L of oxygen. This volume can provide about 20 hours of breathable air for four people.
An oxygen candle cannot have its oxygen production regulated, and once ignited, it will produce oxygen at a rate of approx. 28 Litres per minute until it is exhausted.
Oxygen candles inside a refuge chamber are considered a last resort, only ignited when both compressed air and oxygen cylinders are depleted.

Air Quality Standards for Refuge Chambers
Within Australia and New Zealand, to provide a true source of breathable air, a refuge chamber’s filtration system must comply with AS/NZS 1716:2003 and clause 9.16 from the air supply standard AS/NZS 1715:2009 or their international equivalents.
This standard states that the air supplied to the refuge chambers shall:
- Have no objectionable or nauseating odour; and
- Contain no less than 19.5% and not more than 22% by volume of oxygen.
Additionally, at 15°C and 100 kPa absolute, the air must:
- Contain no more than 11 mg/m3 (10 p.p.m. by volume) of carbon monoxide;
- Contain no more than 1400 mg/m3 (800 p.p.m. by volume) of carbon dioxide;
- Contain no more than 1 mg/m3 of oil, and
- Regular testing of the air at the respirator should be undertaken to verify the quality of the air, with records kept of these tests.
Meeting the standard is generally not attainable to an untreated compressed air supply due to the compressors’ nature, location, and plumbing network. As such, air treatment and secondary breathable air sources are required to maintain a constant supply of safe, breathable air for refuge chamber occupants in an emergency.







